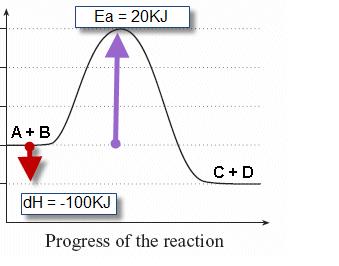
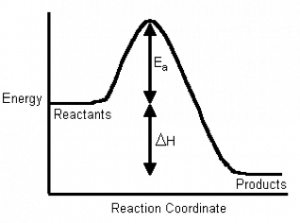
45 label the following reaction energy diagram for a catalyzed and an uncatalyzed process
Solved Label the following reaction energy diagram for a Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Uncatalyzed Catalyzed Eare) with catalyst Ea{fwd) with ... Solved Label the following reaction energy diagram for a Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Reactants State Products Catalyzed Uncatalyzed | | ΔHnn ...
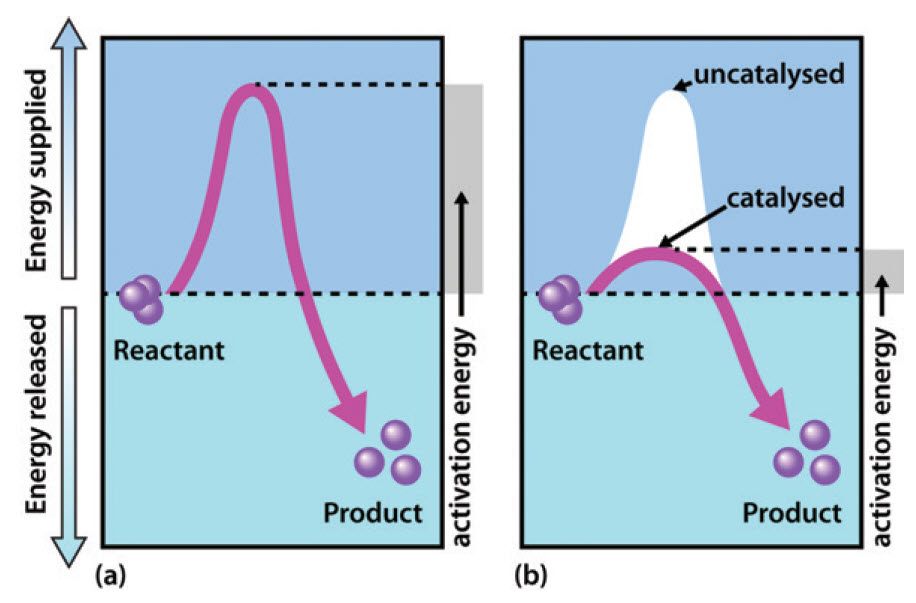
Potential Energy Diagrams - Kentchemistry.com (1) A catalyst is added as an additional reactant and is consumed but not regenerated. (2) A catalyst limits the amount of reactants used. (3) A catalyst changes the kinds of products produced. (4) A catalyst provides an alternate reaction path-way that requires less activation energy. Answer--> choice 4
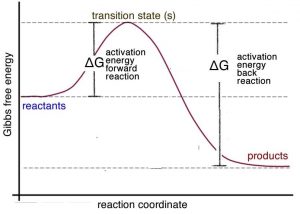
Label the following reaction energy diagram for a catalyzed and an uncatalyzed process
Label the reactants and products on the enthalpy diagrams for each process. Q: At 552.3 k, the rate constant for the thermal decomposition of f so2cl2 is 1.02k2*10^-6. If the activation energy is 210kj/mol determine the rate constant at 700k. Posted 26 days ago. Q: CHM 703 Electro-analytical Techniques 1a. Explain the principle of anodic stripping voltammetry. Why is stripping the most sensitive polarographic technique? PDF The Leon M. Goldstein High School for the Sciences The potential energy diagram below represents a reaction. Reaction Coordínate Which arrow represents the activation energy of the forward reaction? (1) A 2) B 3) C (4) D 5. Which phase changeis anexothermic process? (2) NH3 (g) (1) cu(s) cu(l) (4) Hg(l) Hg(g) 16. Sketch the potential energy diagram for an endothermic chemical reaction that ... Chapter 8 Flashcards | Quizlet The enzyme urease catalyzes the hydrolysis of urea to ammonia plus carbon dioxide. At 21 C the uncatalyzed reaction has an activation energy of about 125 kJ/mol , whereas in the presence of urease the activation energy is lowered to about 46 kJ/mol . By what factor does urease increase the velocity of the reaction? 1.1 x 10^14
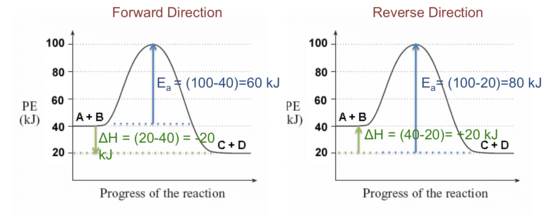
Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Chapter 8 BIOCHEM Flashcards | Quizlet Label the energy reaction graph for the following reaction showing the energy profile for a catalyzed and an uncatalyzed reaction. Ea, rxn not cat by enzyme, rxn cat by enzyme, energy released by rxn A.Look at the graph of reaction rate versus substrate concentration for an enzyme.In which region does the reaction rate remain constant? B. Solved Label the following reaction energy diagram for a Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. AHx>0 AH 0 Eartwd) with catalyst Reaction Intermediate ... Solved Label the following reaction energy diagram for a - Chegg Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. A > 0 Eatwa) no catalyst Exter, with catalyst Reactants Esinev) no Eastw with catalyst Potential energy Mono Products Uncatalyzed Catalyzed Transition State Reaction Intermediate Reaction progress Reset Zoom This problem has been solved! See the answer [Solved] Suzuki Post-Lab 1. Safety Question Define PEL ... - Course Hero For a catalyzed process assume that the rate determining step is the transmetallation of the complex analogous to Complex A to produce the complex analogous to Complex B in PowerPoint slide 18 Label the following in your potential energy diagram: i. Reactants ii. Product iii. AH* for the uncatalyzed process and value given. iv .
Solved Label the following reaction energy diagram for a Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Arx > 0 Transition Stato Reactants Entwu no catalyst ... PDF Forest Hills High School A potential energy diagram is shown below. thout Cataìyst Catalyst Reaction Coordinate Which reaction would have the lowest activation energy? A the forward catalyzed reaction B) the forward uncatalyzed reaction C) the reverse catalyzed reaction D) the reverse uncatalyzed reaction 15. Which conditions will increase the rate of a chemical reaction? PDF Chemical kinetics Name: Date On the diagram below, draw a potential energy diagram for this reaction. 18. Base your answer(s) to the following question(s) on the reaction represented by the balanced equation below. 2H2(g)+O2(g) !2H2O(')+571:6kJ On the axes below, draw a potential energy diagram for the reaction represented by this equation. 19. Given the reaction at ... PDF Graph Review Wed - North St. Paul-Maplewood Oakdale / Overview Below is an energy diagram for a particular process. One curve represents the energy profile for the uncatalyzed reaction, and the other curve represents the energy profile for the catalyzed reaction. Course reaction a. N%ich curve has the geater activation energy? Curve 1 or cunre 2? b.
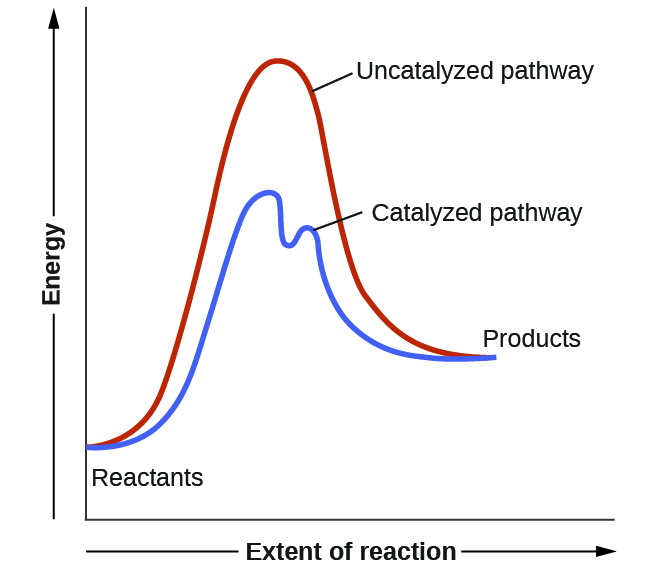
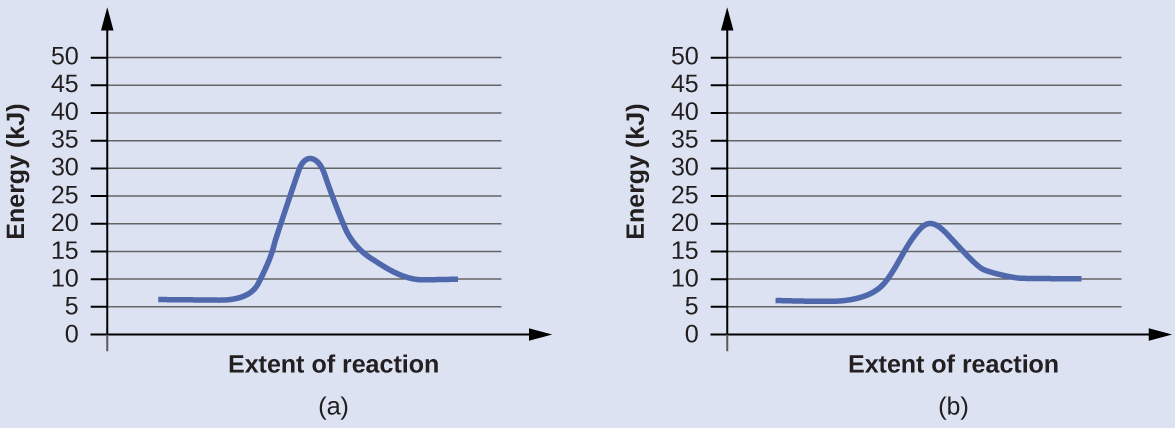
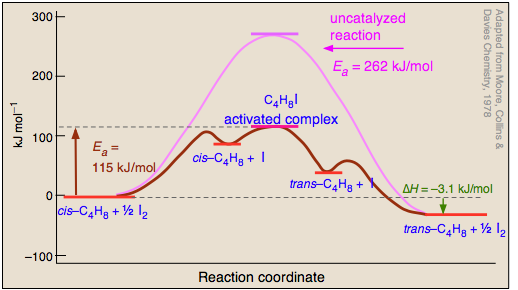
Reaction profiles - Exothermic and endothermic reactions - AQA - BBC Reaction profiles An energy level diagram shows whether a reaction is exothermic or endothermic. It shows the energy in the reactants and products, and the difference in energy between them.... Answered: Sketch a qualitative reaction energy… | bartleby Sketch a qualitative reaction energy diagram for a chemical reaction with and without a catalyst. Assume the uncatalyzed reaction is endothermic. Note: Because the sketches are only qualitative, the energies in them don't have to be exact. They only have to have the right relationship to each other. 12.7 Catalysis - Chemistry - opentextbc.ca A comparison of the reaction coordinate diagrams (also known as energy diagrams) for catalyzed and uncatalyzed alkene hydrogenation is shown in Figure 1. Figure 1. This graph compares the reaction coordinates for catalyzed and uncatalyzed alkene hydrogenation. Solved Label the following reaction energy diagram for a | Chegg.com Transcribed image text: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Arx > 0 Transition Stato Reactants Entwu no ...
PDF i.e. Useful equations and schemes are given on a tear-off sheet ... - UMD (c; 9 pts) The rate-limiting step of the uncatalyzed reaction is the same chemical transformation as for the catalyzed reaction, at least around pH 7. Given this fact, on the diagram below sketch in the remainder of the free energy reaction coordinate diagram for the uncatalyzed reaction. Briefly explain your reasoning.
Solved Label the following reaction energy diagram for a Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. AHpXn > 0 Ealfwd) no catalyst Earrev) with catalyst ...
PDF AP CHEMISTRY 2006 SCORING GUIDELINES - College Board (d) Consider the four reaction-energy profile diagrams shown below. (i) Identify the two diagrams that could represent a catalyzed and an uncatalyzed reaction pathway for the same reaction. Indicate which of the two diagrams represents the catalyzed reaction pathway for the reaction. Diagram 1 represents a catalyzed pathway and diagram 2
Label the following reaction energy diagram for a catalyzed and an ... SOMEONE ASKED 👇. Label the following reaction energy diagram for a catalyzed and an uncatalyzed process.
Answered: Label the components of an energy… | bartleby Label the components of an energy diagram for a spontaneous reaction. Question Transcribed Image Text: Label the components of an energy diagram for a spontaneous reaction. 4 Energy- Reaction progress- Answer Bank activation energy catalyzed reaction reactants products uncatalyzed reaction
Solved Label the following reaction energy diagram for a Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process Transition State Reactants Products Catalyzed Uncatalyzed ...
Energy diagrams for enzyme‐catalyzed reactions: Concepts and ... - IUBMB The energy diagram for a reaction model consisting of one enzyme, one substrate, and one product is depicted in many books where it is compared with that for the uncatalyzed reaction. The survey of several Biochemistry textbooks reveals a high diversity of profiles for the same process. A, C, and D are adapted from Refs. 5, 3, and 2, respectively.
The graph that best describes catalyzed and uncatalyzed exothermic ... In the presence of a palladium catalyst, the ethyne forms ethene and ethane. This reaction is represented by the unbalanced equation C2H2(g) + H2(g) C2H4(g) + C2H6(g) + energy The energy diagram that represents both the catalyzed (---) and uncatalyzed reactions (___) is a. c. b. d. Upload your study docs or become a
Energy Changes in Catalytic Reactions - QS Study The catalyzed reaction is shown as a two-stage process with lower activation energy and is, therefore, faster. It is possible that the unanalyzed reaction proceeds by more than one step and the catalyzed reaction may also consist of several steps. The important point to note is that for the catalyzed reaction there is no step which has higher ...
PDF Potential Energy Diagram Worksheet ANSWERS A catalyst changes the reaction mechanism, in the process lowering the activation energy. 5. Name 4 things that will speed up or slow down a chemical reaction. Increase concentration by distillation of a solvent, Increase concentration by increasing pressure of a gas, Increase temp, Add a catalyst, Add an inhibitor. 6. Draw an energy diagram ...
Solved Label the following reaction energy diagram for a - Chegg Expert Answer 100% (64 ratings) All the labeling you have done are correct except Hrxn < 0. Hrxn<0 because i … View the full answer Transcribed image text: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process.
Solved Label the following reaction energy diagram for a Image for Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. help!! thx. Show transcribed image text ...
PDF ap07 chemistry q6 - College Board The reaction is exothermic. The reaction is slow at 25°C; however, a catalyst will cause the reaction to proceed faster. (e) Using the axes provided below, draw the complete potential-energy diagram for both the catalyzed and uncatalyzed reactions. Clearly label the curve that represents the catalyzed reaction.
How to draw the potential energy diagram for this reaction? C3H8(g) + 5O2(g) → 3CO2(g) +4H2O(g) + 2219.9 kJ, we say that ΔH ∘ C = − 2219.9 kJ/mol propane. We approximate that this is the change in potential energy for the reactants going to the products. The above is for an endothermic reaction.
PDF Energy/Reaction Coordinate Diagrams - chemconnections A Reaction Coordinate (Energy) Diagram Thermodynamic Quantities Gibbs standard free energy change ... Bonds are NOT in the process of breaking or forming. ! Energy Diagrams # Intermediates! 6! For each of the diagrams below, will the transition state ... Label each of those atoms nucleophilic (nu) or electrophilic (el) in each resonance ...
Chapter 8 Flashcards | Quizlet The enzyme urease catalyzes the hydrolysis of urea to ammonia plus carbon dioxide. At 21 C the uncatalyzed reaction has an activation energy of about 125 kJ/mol , whereas in the presence of urease the activation energy is lowered to about 46 kJ/mol . By what factor does urease increase the velocity of the reaction? 1.1 x 10^14
PDF The Leon M. Goldstein High School for the Sciences The potential energy diagram below represents a reaction. Reaction Coordínate Which arrow represents the activation energy of the forward reaction? (1) A 2) B 3) C (4) D 5. Which phase changeis anexothermic process? (2) NH3 (g) (1) cu(s) cu(l) (4) Hg(l) Hg(g) 16. Sketch the potential energy diagram for an endothermic chemical reaction that ...
Label the reactants and products on the enthalpy diagrams for each process. Q: At 552.3 k, the rate constant for the thermal decomposition of f so2cl2 is 1.02k2*10^-6. If the activation energy is 210kj/mol determine the rate constant at 700k. Posted 26 days ago. Q: CHM 703 Electro-analytical Techniques 1a. Explain the principle of anodic stripping voltammetry. Why is stripping the most sensitive polarographic technique?
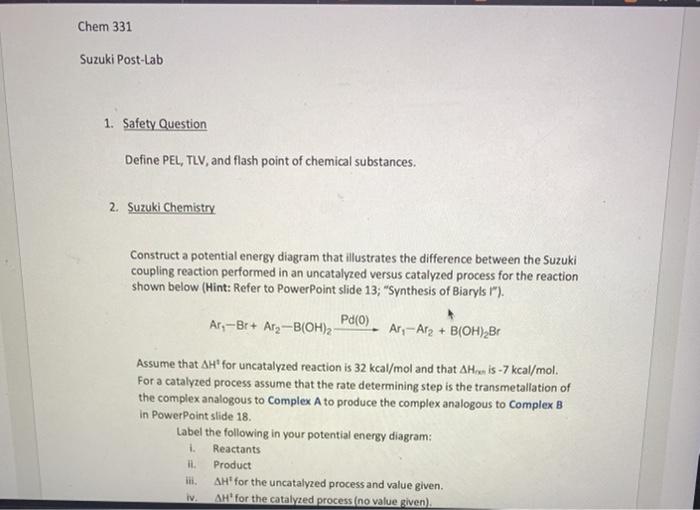
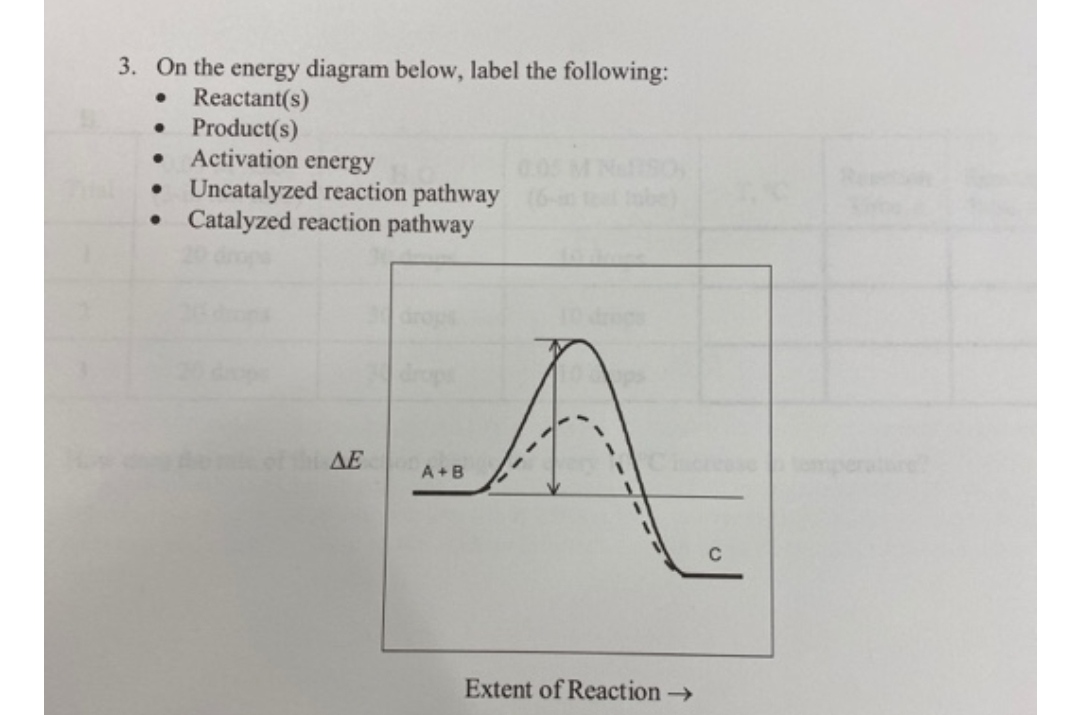








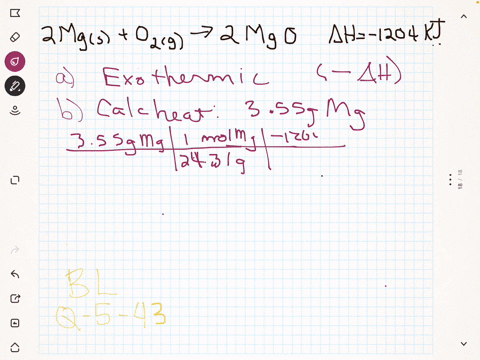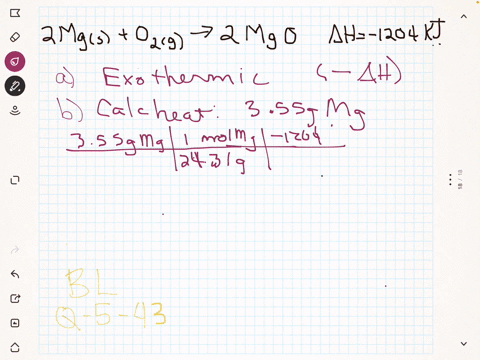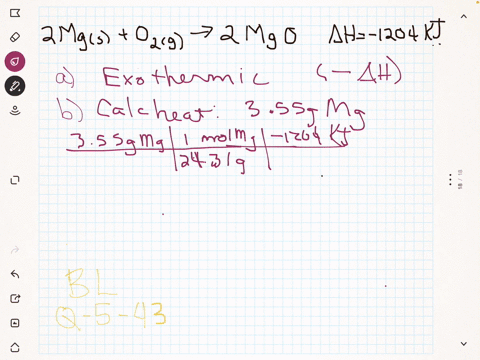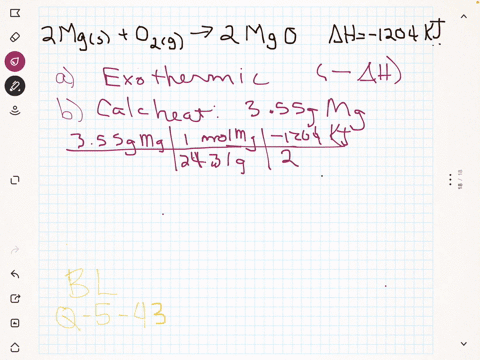







Post a Comment for "45 label the following reaction energy diagram for a catalyzed and an uncatalyzed process"